entimICE FastTrack Statistical Computing Environment significantly increases the efficiency of the clinical data science processes, managing constantly growing study pipelines, resource bottlenecks, and ever-increasing regulatory requirements. entimICE FastTrack is ready today for data from decentralized clinical trials and various sources, and fully supports real-time analysis and reporting along with the use of AI/ML.
Solutions for every customer

Accelerated Time to Market
entimICE FastTrack significantly increases the efficiency of clinical data science processes, managing growing study pipelines and resource bottlenecks t

Accelerated Time to Market
entimICE FastTrack significantly increases the efficiency of clinical data science processes, managing growing study pipelines and resource bottlenecks t

Accelerated Time to Market
entimICE FastTrack significantly increases the efficiency of clinical data science processes, managing growing study pipelines and resource bottlenecks t

Accelerated Time to Market
entimICE FastTrack significantly increases the efficiency of clinical data science processes, managing growing study pipelines and resource bottlenecks t

Accelerated Time to Market
entimICE FastTrack significantly increases the efficiency of clinical data science processes, managing growing study pipelines and resource bottlenecks t

Accelerated Time to Market
entimICE FastTrack significantly increases the efficiency of clinical data science processes, managing growing study pipelines and resource bottlenecks t
Solutions for every customer
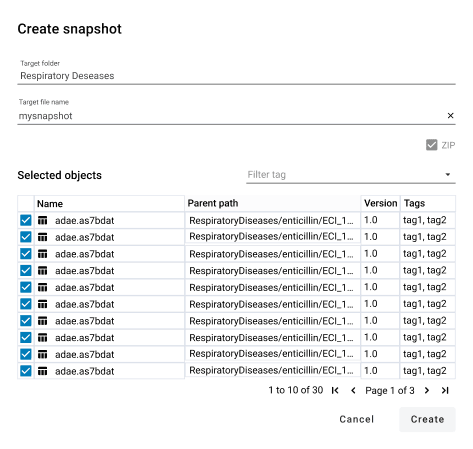
Statistical Computing Environment
- Language-agnostic development with direct access to native SAS, Posit (RStudio), and Python environments - enabling users to code in their preferred interface without leaving the platform.
- Automatic traceability of program executions and clinical data flow, enabling the viewing of 'bigger picture' throgh interactive graphs and automated re-runs for outdated outputs.
- Inbuild robust workflows from development to production facilitating lifecycle management and validation, ensuring quality control, documentation and signature driven approval.
- Automation tools, including program run sequences and job manager which eliminate repetitive tasks. Our open API enables integration and automation across external systems and enterprise workflows.
- Configurable non-blocking interface to provide optimal support for individual working styles. Flexible dashboards guide users to the repository objects.

Universal Data Access
- Access any data source including clinical, legacy, biomarker, lab, device and real-world data to utilise your study data quickly and easily.
- Open REST APIs enable connectivity with upstream data sources for controlled access to data and metadata in entimICE FastTrack.
- Secure role based access to data to ensure data security and compliance.
- Ingest structured and unstructured data in real time with automated quality checks.
- Manage data standards centrally and apply them at global or study levels.
- Intuitive search and navigation features, so that users can quickly find artefacts of interest such as programs, datasets, or output files.

Clinical Data Science Ecosystem
- entimICE FastTrack is a cloud native, integrated clinical platform that combines the power of a Statistical Computing Environment, open source tools and out-of-the box workflows to stream analysis and reporting activities.
- Based on microservices with significantly shorter test and validation cycles, enabling scalability for business growth.
- Standardised and validated REST APIs allow easy integration of any third party commercial or open-source tool with entimICE FastTrack.
- Enhanced study specific metadata and terminology support to enhance data standardisation, validation and review processes.

Enterprise Collaboration
- Study Progress dashboards and reports provide a complete overview of activities in all ongoing studies and enables active task and deliverable management.
- Built-in workflows ensure the process management of all objects covering lifecycle management and QC framework for submission review.
- Issue tracking at all levels with automatic notifications with full audit trail enable faster query resolution, eliminating the need of traditional and tedious spreadsheet based tracking.
- Collaborate with study teams to resolve programming and source data discrepancies and medical writers to streamline clinical data review.

Security and Compliance
- Automatic end-to-end traceability and effortless compliance with regulatory requirements, including FDA 21 CFR Part 11, EU Annex 11 and ICH GxP.
- Flexible search and reporting engine so you can easily find relevant information from version control, audit trails, object history and execution traces.
- Flexible role based permission model that enables team to manage access control for internal users and partners with confidence.
- Reliable and reportable electronics signatures to validate relevant workflows.
- Comprehensive, tamper-proof audit trails that log every action, change, and decision - ensuring full accountability, transparency, and inspection readiness.
Solutions for every customer

Efficient and GxP compliant clinical trial analysis and reporting
ectrum. In addition toxicokinetic studies for non-individualized (rodents) and individualized animals are supported. PhaLIMS includes a set of integrated administration, management, planning, proces

Efficient and GxP compliant clinical trial analysis and reporting
ectrum. In addition toxicokinetic studies for non-individualized (rodents) and individualized animals are supported. PhaLIMS includes a set of integrated administration, management, planning, proces

Efficient and GxP compliant clinical trial analysis and reporting
ectrum. In addition toxicokinetic studies for non-individualized (rodents) and individualized animals are supported. PhaLIMS includes a set of integrated administration, management, planning, proces

Efficient and GxP compliant clinical trial analysis and reporting
ectrum. In addition toxicokinetic studies for non-individualized (rodents) and individualized animals are supported. PhaLIMS includes a set of integrated administration, management, planning, proces

Efficient and GxP compliant clinical trial analysis and reporting
ectrum. In addition toxicokinetic studies for non-individualized (rodents) and individualized animals are supported. PhaLIMS includes a set of integrated administration, management, planning, proces
- Efficient and GxP compliant clinical trial analysis and reporting
- Efficient and GxP compliant clinical trial analysis and reporting
- Efficient and GxP compliant clinical trial analysis and reporting
- Efficient and GxP compliant clinical trial analysis and reporting
- Efficient and GxP compliant clinical trial analysis and reporting